|
Mårtensson, "Core-Level Binding Energies in Metals," J. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Electronic configuration: It is the arrangement of electrons in the increasing order of energy in the shells of an atom. electron spin residing on the cobalt(II) atom with the 59Co nuclear spin (I. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. cobalt(II) atom of AdoCbl having low-spin d7electronic configuration and the. 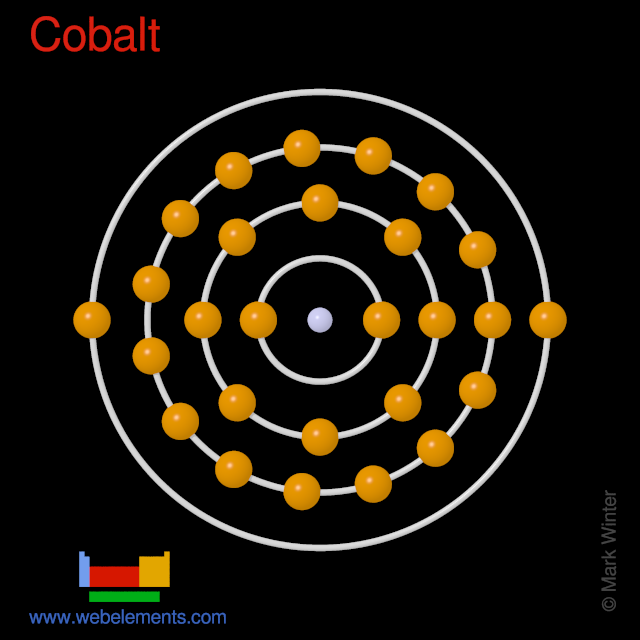
In this state, if we see the electron configuration of Cobalt, then it possesses incomplete d-orbitals. And the ground state electronic configuration of Cobalt is Ar 4s 2 3d 7. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). Now, the ground state of Cobalt means its normal state in which it has neither gained nor lost any electron/s. The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. All values of electron binding energies are given in eV. 1967, 47, 1300.Įlectron binding energies Electron binding energies for cobalt. Only three quantum numbers are needed to uniquely describe an electron. In the buildup of atoms, electrons occupy the 4 f orbitals before the 6 s orbitals. This electron configuration allows the 3 single electrons to all spin the same direction creating an electrical current which causes Cobalt to be one of the naturally magnetic elements. Two of the three d orbitals have two electrons. Ni Ni has two unpaired electrons in its 3 d orbitals. The last 7 electrons go into the five 3 d orbitals. These effective nuclear charges, Z eff, are adapted from the following references: An electron in a 2 s orbital can have the same n, l, and ml quantum numbers as an electron in a 3 s orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
